Shilatifard Laboratory
Recent Videos from the Shilatifard Lab
News from the Shilatifard Lab
What We Do
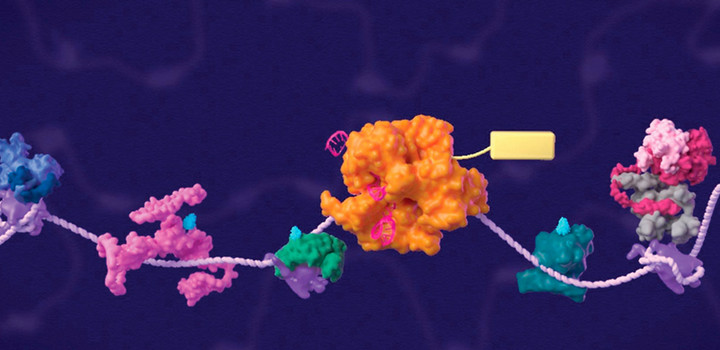
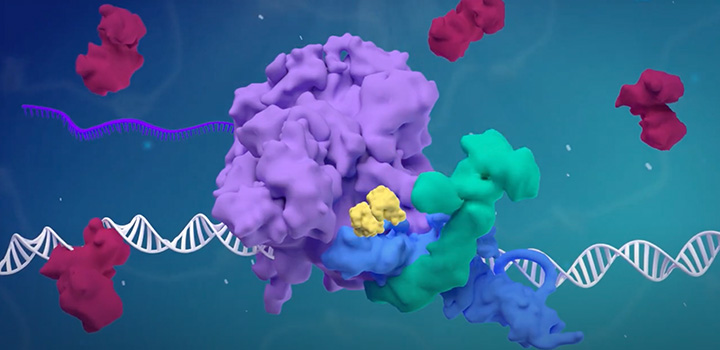
The regulation of gene expression by RNA polymerase II is essential for development and differentiation, and its misregulation contributes to the pathogenesis of many forms of cancers. The goal of our laboratory is to define the molecular mechanisms underlying cancer pathogenesis and to identify potential targets for therapy. These studies have been done through detailed understanding of proteins and protein complexes that regulate chromatin modifications, transcriptional initiation, and transcriptional elongation. Several of such findings from our lab are now under clinical studies for the treatment of bladder cancer and pediatric brain tumors.
Taking A Northwestern Direction
“Northwestern isn’t just a compass point on a map. Here, it’s a mindset. A Northwestern Direction is a journey that each one of us creates and defines as our own. There’s always a new adventure, a new idea, and a new discovery—no matter what path you pursue.”