Research
The research efforts of the Laimins Laboratory are focused on three main areas:
Pathogenesis of papillomavirus in differentiating epithelia

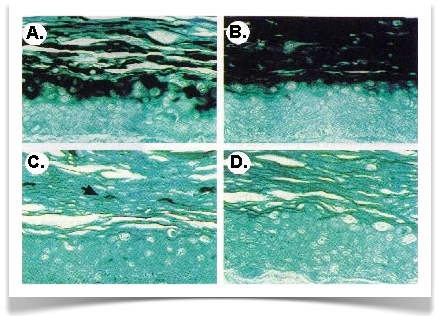
The papillomavirus life cycle is closely linked to epithelial differentiation and progeny virus are replicated in terminally differentiated epithelial cells. We have used organotypic tissue culture systems to faithfully reproduce the differentiation program of epithelial cells in the laboratory and to generate papillomavirus virions (Fig. 1). Recently, we have developed methods to genetically analyze HPV functions during the viral life cycle. Current efforts are directed at analyzing the functions of viral early proteins in the life cycle. Over fifty different mutant viral genomes have been examined to date.
The role of the viral oncoproteins, E6 and E7 in oncogenesis
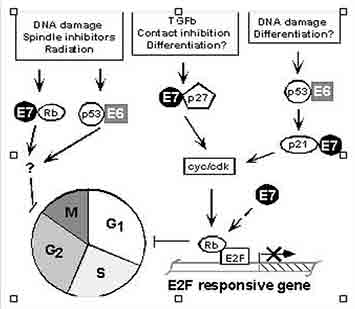
Figure 2. The viral oncoprotein E6 and E7 interact with numerous cellular proteins disrupting regulation of the cell cycle.
The regulation of viral gene expression
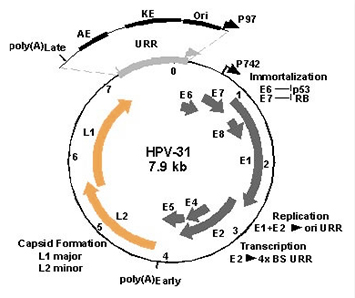
Figure 3. The dsDNA circular genome of HPV type 31. Viral gene expression is regulated by a keratinocyte specific enhancer (KE) and an auxilary enhancer (AE) located within the upstream regulatory region (URR). Early transcripts initiate at the promoter P97 while late transcripts initiate at the promoter P742 which is activated upon keratinocyte differentiation. Early transcripts utilize a polyadenylation signal (poly(A)Early) downstream of the E5 open reading frame while late transcripts use the signal (poly(A)Late) downstream of the L1 open reading frame.
The regulation of viral gene expression occurs at both the initiation of transcription and through post-transcriptional mechanism (Fig. 3). On going studies are examining the transcription factors that regulate early gene expression in undifferentiated cells and late gene expression in differentiated cells. Gene expression is also regulated through post-transcriptional control of splicing and polyadenylation. A set of tandem polyadenylation signals allows the virus to discriminate between expression of early and late genes. Efforts are in progress to dissect the mechanisms regulating this switch in polyadenylation site usage.